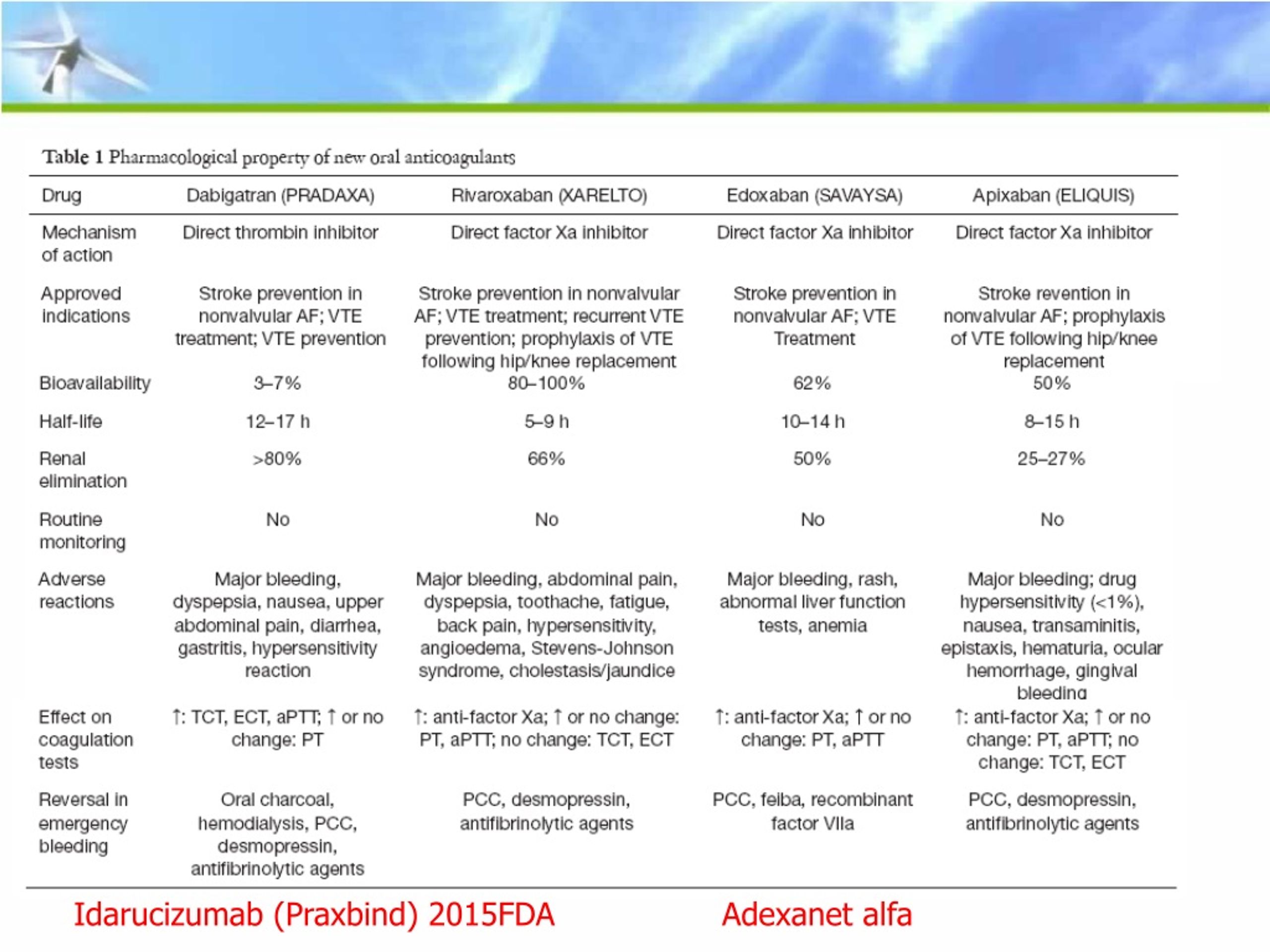
Praxbind is the first reversal agent approved specifically for Pradaxa and works by binding to the drug compound to neutralize its effect, the FDA said. The FDA approved Pradaxa in 2010 to prevent stroke and systemic blood clots in patients with atrial fibrillation, as well as for the treatment and prevention of deep venous thrombosis and pulmonary embolism. “Today’s approval offers the medical community an important tool for managing patients taking Pradaxa in emergency or life-threatening situations when bleeding can’t be controlled,” he added. “The anticoagulant effects of Pradaxa are important and life-saving for some patients, but there are situations where reversal of the drug’s effects is medically necessary,” Richard Pazdur, MD, director of the Office of Hematology and Oncology Products in the FDA’s Center for Drug Evaluation and Research, said in a statement. Indivior, Drug to Fight Opioid Addiction, Approved by U.S.Subscribe to newsletters for the latest medication news, new drug approvals, alerts and updates. FDA Approves First-Ever Implant to Treat Opioid Addiction PRAXBIND treatment can be used in conjunction with standard supportive measures, which should be considered as medically appropriate see Clinical Pharmacology. 
Blood-Thinner Reversal Agent Works in 82% of Serious Bleeding Cases One study sponsored by the manufacturer found that idarucizumab effectively reversed anticoagulation caused by dabigatran within minutes.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
